Coupa for Pharma vs Hyperbots: Regulated Procurement & Supplier Compliance Compared
In regulated pharma, procurement control isn’t enough. This comparison shows why invoice accuracy, CFR-ready traceability, and GL-confirmed posting define true compliance.

Pharma procurement is a little like a lab experiment: a single mis-measured ingredient can ruin a batch and sometimes a regulatory filing. When you’re buying temperature-sensitive reagents, implanted devices, or controlled compounds, procurement isn’t just procurement. It’s traceability, CFR-friendly evidence, supplier validation, and the ability to prove, in forensic detail, what happened and when. That’s why choosing tools matters.
Coupa for pharmaceuticals is a broad, powerfully opinionated source-to-pay control tower but how does it compare with Hyperbots, an invoice-first, GL-centric automation engine? The short version: Coupa is good at governance; Hyperbots is built to make the books auditable, fast, and boring (the good kind of boring).
Where Coupa wins and why pharma teams install it in the first place
Coupa is marketed as a total spend management platform with explicit industry modules for life sciences and healthcare. It centralizes sourcing, contracting, supplier portals, and analytics, all useful for organizations that must enforce preferred supplier programs, control maverick spend, and surface savings at scale. Many pharma and healthcare organizations deploy Coupa for exactly that reason.
Typical procurement wins with Coupa:
Centralized catalogs and PO governance that reduce off-contract purchases (a practical way to protect thin margins).
Supplier portals to onboard vendors and standardize communications (good for supplier management and visibility).
Source-to-contract capabilities that help enforce negotiated pricing and supplier performance monitoring across global operations.
If your priority is enterprise-grade sourcing and supplier governance across a global pharma footprint, Coupa is a sensible, widely-adopted choice. Think of it as the control tower that keeps airplanes from colliding: essential, authoritative, and excellent at preventing unauthorized flights.
Where Coupa limitations in pharma compliance start to matter
But procurement governance is only half the story in regulated pharma. The other half is financial traceability: can you prove that every lot, every certificate of analysis (COA), every GxP attestation and invoice line ended up in the ledger with the right metadata? That’s where some Coupa deployments require extra gymnastics.
Common pain points in pharma implementations:
Supplier validation challenges: Pharma onboarding requires CFR-style evidence (supplier quality agreements, COAs, regulatory attestations).
Suppliers upload heterogeneous documents, PDFs, scanned certificates, Word docs, and those artifacts don’t always map cleanly into downstream financial records.
Coupa’s supplier portal centralizes registration and communications, but the documentation ecosystem remains varied and often lives in attachments.
CFR documentation overhead: Regulatory provenance (lot/serial, COA links, chain-of-custody artifacts) must be preserved through to the journal entry.
In many Coupa+ERP architectures, that level of enrichment is performed post-export, in middleware or the ERP, which adds steps, delay, and failure modes.
GL fidelity gaps from integration choreography: Coupa often acts as the source system that exports POs, receipts, and invoices for downstream processing and journal creation.
The “last mile” attaching regulatory metadata and posting audit-ready journals usually happens outside Coupa, which increases the places where things can go wrong.
In short, Coupa reduces procurement risk; it doesn’t automatically guarantee day-one, audit-ready GL entries for regulated goods. In pharma that gap can mean exceptions, reconciliations, and potential findings in an audit which is the exact opposite of a quiet month-end.
Why GL posting and invoice accuracy are mission-critical in pharma
A few reasons this matters more in pharma than in many other industries:
Regulatory traceability: Lot numbers, GTIN/UDI/serials, COAs and supplier qualifications must be traceable to payments and journals for FDA/EMA audits. Any break in the chain triggers remediation work.
Accrual precision: Pharma uses accrual accounting for expensive consumables and capitalized equipment; late or incorrect postings distort financial statements and budgeting.
High consequence of errors: A misposted invoice for temperature-controlled shipments or implantables can cascade into compliance and inventory reconciliation headaches.
Operational decisioning: Accurate GLs inform procurement cadence for clinical trials, inventory levels for cold-chain materials, and capital planning, wrong numbers lead to wrong operational choices.
In short: in pharma, “close-week calm” is not a nice-to-have, it’s a compliance imperative.
Enter Hyperbots: invoice-first, GL-centric, and built for accuracy
Hyperbots takes a different, purpose-built approach: treat the invoice as the transaction discover it, extract it, enrich it, validate it, and post a confirmed journal back to the ERP. That pipeline matters because it collapses the hops that generate exceptions.
What Hyperbots brings to pharma finance (claims supported by Hyperbots materials):
Near-perfect extraction (~99.8%) for field-level invoice data, including multi-page, complex invoices. This reduces extraction errors that would otherwise cause manual queues.
Line-item and tabular parsing that preserves multi-line structure (so a 12-page surgical kit invoice isn’t flattened into garbage).
End-to-end automation (discovery → extraction → match → GL posting) and ERP read-back validation so journals are posted and confirmed, not assumed. Hyperbots describes read-back checks and retries as part of its connector approach.
High straight-through processing (STP ~80%), meaning the majority of invoices flow without human touch dramatically reducing recurring manual effort.
Hyperbots’ philosophy is pragmatic: don’t build every module; build the part that stops exceptions and protects the ledger. For pharma, that’s a huge win.
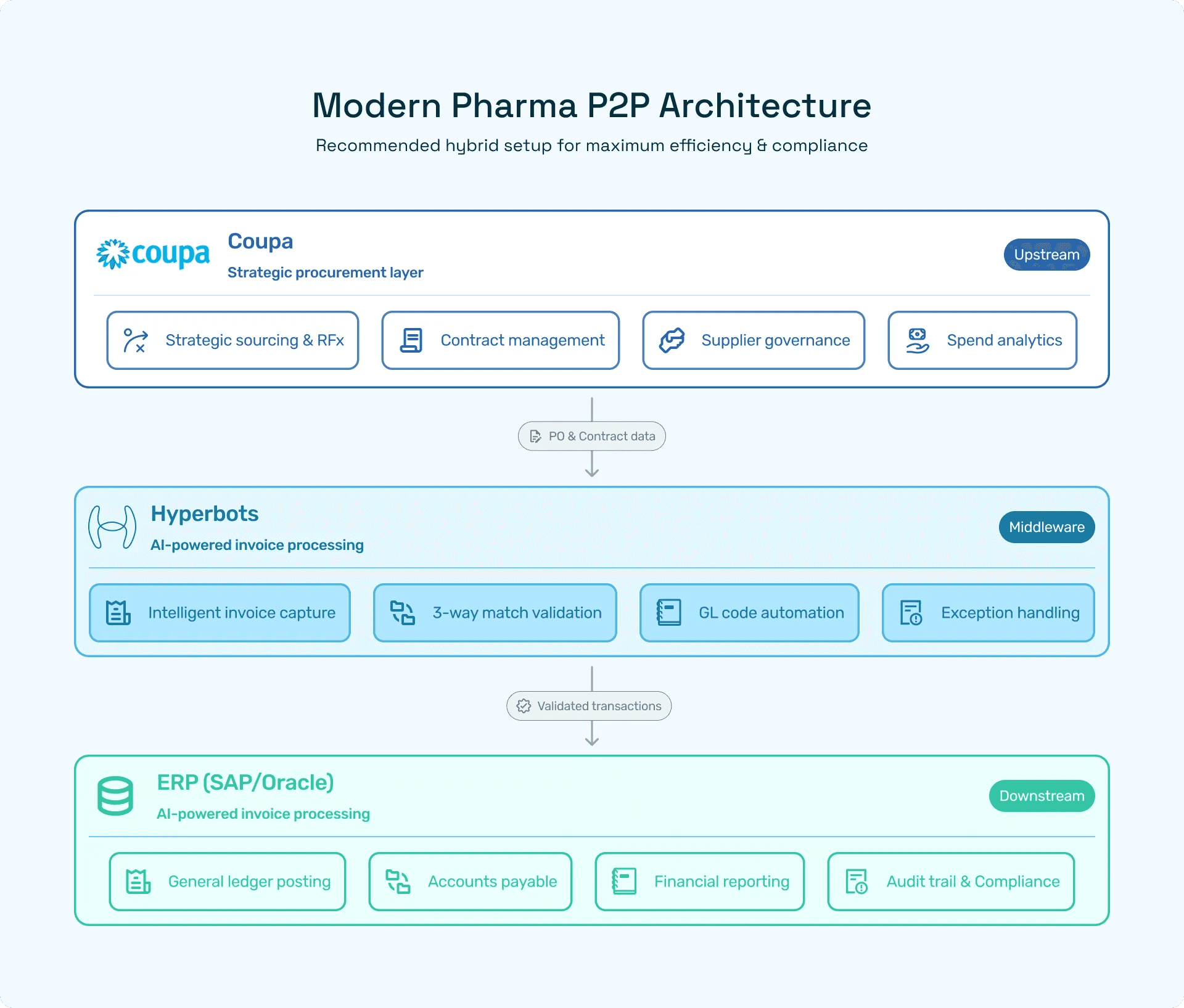
The hybrid playbook (how smart pharma teams actually deploy tools)
This is rarely an either/or choice. The effective, risk-aware architecture in regulated pharma is usually hybrid:
Coupa for sourcing & supplier governance: keep the control tower for contracts, preferred-vendor programs, and cross-enterprise procurement policy enforcement.
Hyperbots for invoice capture & GL fidelity: layer an invoice-first engine to guarantee the ledger is accurate, auditable, and posted with read-back confirmation.
That combo protects both the strategic procurement agenda and the financial audit posture. Procurement keeps negotiating savings; finance keeps the books pristine.
Final verdict: compliance-savvy procurement + GL-obsessed finance = win
Coupa for pharmaceuticals is a market-leading procurement platform: essential for sourcing, contracts, and supplier governance. But when the audit team asks for traceability, lot numbers tied to journals, COAs linked to payments, and accruals posted with SOX-grade confirmation, an invoice-first approach wins the day.
Hyperbots’ high extraction accuracy, multi-page line-item parsing, STP, and ERP read-back validation turn the invoice into a reliable, auditable pipeline. For pharma organizations juggling regulatory metadata and life-critical supply chains, the pragmatic architecture is clear:
Keep Coupa for sourcing. Add Hyperbots to make your finances auditable, fast, and boring (which is a very good thing in finance).
Frequently Asked Questions (FAQs)
Q1. Why is invoice accuracy more critical in pharma than in other industries?
Pharma invoices often include regulated metadata such as lot numbers, serials, COAs, and temperature-controlled shipment details. Even small extraction errors can break audit traceability, trigger exceptions, or create compliance findings during FDA or EMA reviews. High invoice accuracy dramatically reduces reconciliation work and regulatory risk.
Q2. Does Coupa fully support CFR-compliant financial traceability on its own?
Coupa is strong at procurement governance and supplier controls, but in many pharma deployments, CFR-level traceability from invoice to GL journal is completed downstream via ERP logic or middleware. That means audit-ready financial records often depend on integration design rather than native Coupa processing.
Q3. How does Hyperbots support regulated pharma audits differently?
Hyperbots treats the invoice as the system of record and preserves regulatory metadata—such as lot numbers and COA references—through extraction, validation, and ERP posting. Its read-back validation confirms that journals are accepted by the ERP, creating a closed-loop, audit-ready trail from document to ledger.
Q4. Can pharma companies use Coupa and Hyperbots together?
Yes. Many regulated organizations use Coupa for sourcing, contracts, and supplier governance while deploying Hyperbots for invoice accuracy, accrual posting, and GL validation. This hybrid model balances procurement control with finance-grade compliance and faster closes.
Q5. What KPIs should pharma teams track when evaluating AP automation ROI?
Beyond cost per invoice, pharma teams should track exception rate, straight-through processing (STP), time-to-journal posting, audit query volume, and the ability to retain regulatory metadata through the GL. These metrics better reflect compliance risk and sustainable ROI in regulated environments.

